Real Risks of Unregulated Third-Party Servicing
Medical imaging has revolutionized healthcare from the start but especially over the past 30 years. Today, patients and healthcare providers depend on the safe and effective operation of medical imaging devices to diagnose, monitor and guide treatment of cancer and other diseases.
To ensure that patients are receiving safe and accurate scans, the individuals servicing these devices must be highly trained and following “best practices” processes. Accordingly, their employers must be regulated.
Servicing technicians who work for original equipment manufacturers receive extensive training, shadow more experienced technicians, and return for continuing education on new or updated devices – all under the oversight of the Food and Drug Administration (FDA). However, many medical devices are maintained by unregulated third-party servicers who are not required to undergo the same rigorous training program and do not have to register with FDA. This means that some of these highly complicated machines are serviced by businesses that are not required to meet any proper quality and safety Standards. This means their work is done with no oversight.
Without proper oversight, there is an increased risk for serious patient and operator safety and device performance issues. Below are some examples of what happens when medical imaging equipment is improperly serviced by unregulated third parties:
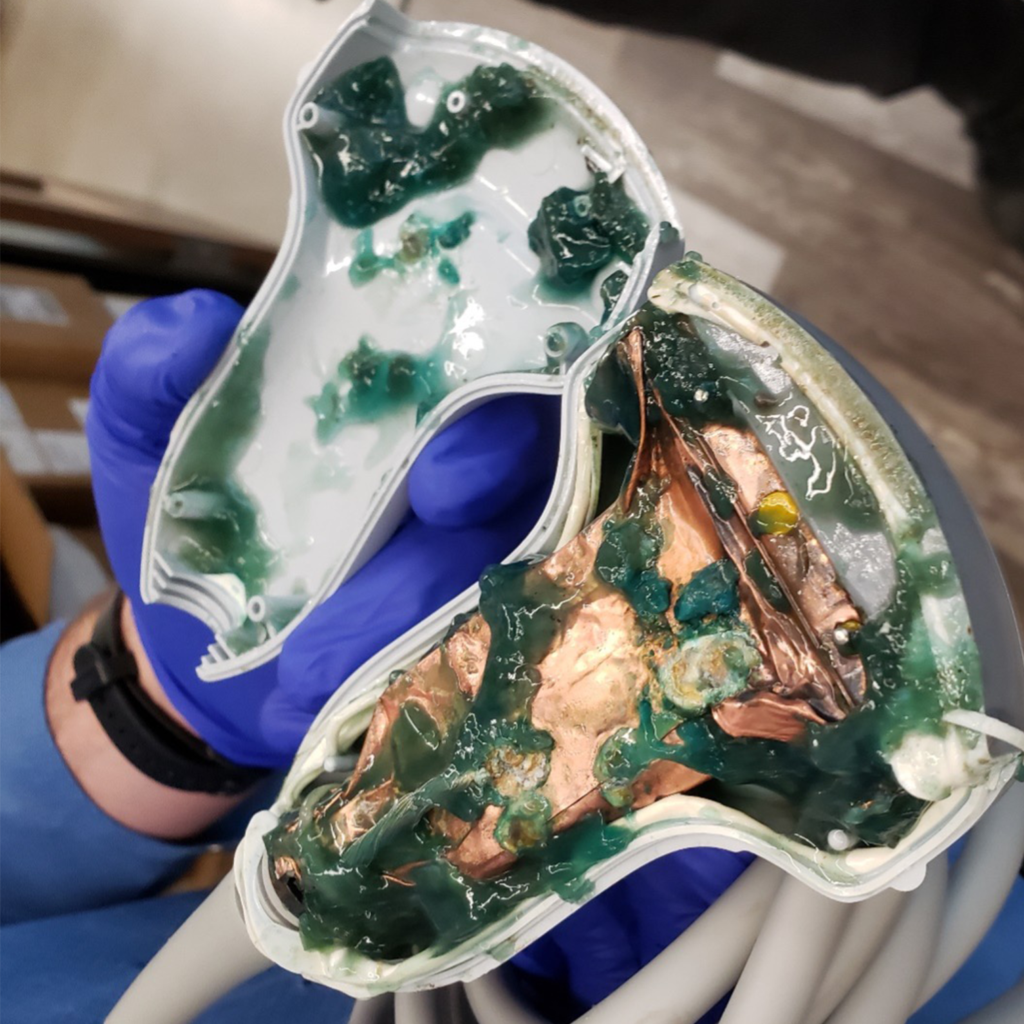
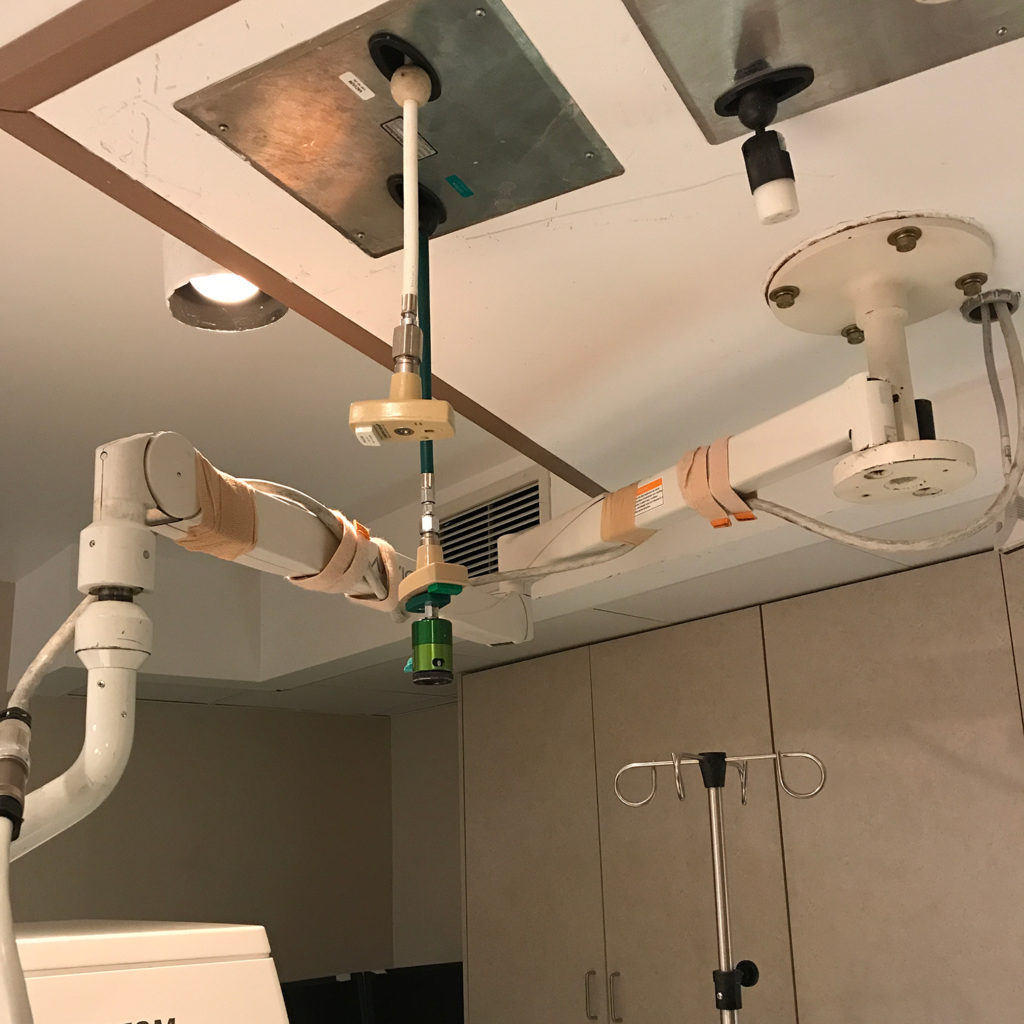


Proper oversight by FDA can help prevent these dangerous situations. FDA should hold all servicing businesses to the same quality, safety, and regulatory requirements, including proper training, implementation of quality and safety controls, adverse event reporting, and registration, to ensure that patients are receiving safe and accurate scans.
More Articles
Our New Year’s Resolution: Ensure Safe Servicing
As we enter the new year, it’s time to set our resolutions for 2022. Here at Right Scan…
Bruce’s Story: A PET Scan Found My Recurrent Prostate Cancer
In 2018, I was diagnosed with prostate cancer. My doctors were able to surgically remove my prostate, and…
Support the FIND Act to Increase Access to Innovative Diagnostics
Through exciting innovations in diagnostic radiopharmaceuticals, doctors are finding new ways to diagnose and monitor conditions such as…
Real Risks of Unregulated Third-Party Servicing
Medical imaging has revolutionized healthcare from the start but especially over the past 30 years. Today, patients and…
